Haberman Associates in “Pink Sheet” article on the cystic fibrosis drug market
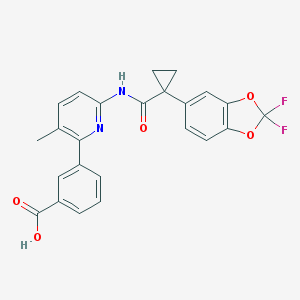
Lumacaftor (Vertex’ VX-809)
I was quoted in an article in the March 11, 2013 issue of Elsevier Business Intelligence’s The Pink Sheet by senior writer Joseph Haas. The article is entitled . A subscription is required to view the full text of this article.
The article focused on the newly-approved disease modifying drug ivacaftor (Vertex’ Kalydeco), as well as programs in drug discovery and development of disease-modifying drugs for cystic fibrosis (CF) at Vertex, PTC Therapeutics, Proteostasis Therapeutics, Pfizer, and Genzyme. It also discussed pipeline products aimed at treating or preventing life-threatening infections in CF patients at such companies as KaloBios, Insmed, and Savara.
Mr. Haas interviewed me for this article. Most of the content of our interview is available in our February 15, 2013 article on the Biopharmconsortium Blog. One company whose R&D program we did not cover in that article is Proteostasis. Proteostasis’ CF program, which is being carried out in collaboration with the Scripps Research Institute, is aimed at discovery and development of compounds that promote CFTR ΔF508 folding and trafficking. This program is in the research and lead optimization stage. We discussed R&D programs at other companies (Vertex, Pfizer) that are also aimed at correction of improper CFTR ΔF508 folding and trafficking in our February 15, 2013 article.
KaloBios’ KB001-A, a bacterial virulence factor-targeting agent
Among the agents aimed at ameliorating life-threatening infections in CF patients that were discussed in the Pink Sheet article is KB001-A, a monoclonal antibody (MAb) agent being developed by KaloBios (South San Francisco, CA). KB001-A is now in Phase 2 development for prevention of Pseudomonas aerguinosa infections in the lungs of CF patients. KB001-A targets an extracellular component of the bacterium’s type III secretion system. This system enables the bacteria to kill immune cells by injection of protein toxins into these cells.
The type III secretion system is an example of a virulence factor. Virulence factors are not expressed by a strain of pathogenic bacteria in vitro, but are expressed only when the bacteria infect a host. Once expressed, they enable the bacteria to colonize the host and cause disease.
In our June 11, 2012 article on this blog, we discussed an antibacterial drug discovery strategy aimed at targeting two related physiological systems that are important in the ability of pathogenic bacteria to cause disease, but are not essential for bacterial proliferation or survival. These systems are virulence factors and quorum sensing. At least by hypothesis, agents that disrupt these systems will prevent pathogenic bacteria from causing disease without selecting for resistant strains of the bacteria. This will give such agents an advantage over conventional antibiotics, which notoriously generate resistant strains when used to treat infections. According to the Pink Sheet article, KaloBios believes that P. aerguinosa bacteria will not develop resistance to KB001-A, which is in accord with this hypothesis.
Another issue with anti-infectives used to treat CF that is discussed in the Pink Sheet article is the definition of a “disease-modifying” agent for CF. We define disease-modifying agents as drugs that ameliorate or cure a disease by targeting the root cause of that disease. However, KaloBios considers KB001-A to be a disease-modifying agent. That is because the company believes that most CF patients die of the effects of P. aerguinosa infection, which causes deterioration of the patients’s lungs. Thus an effective anti-P. aerguinosa agent may produce dramatic increases in patients’ lifespans.
Perhaps the real issue is that one should not classify CF drugs as “disease-modifying” agent and agents that merely treat “symptoms” (as is done in the Pink Sheet article) but should define infections of CF patients as “complications” of the disease. Thus anti-infectives such as KB001-A may effectively treat a major life-threatening complication of CF, without modifying the underlying disease. Such an agent would result in increased lifespans (and improved quality of life) for CF patients, without affecting their underlying disease. As KaloBios asserts, anti-infective agents like KB001-A would be complementary to such disease-modifying agents as ivacaftor.
________________________________
As the producers of this blog, and as consultants to the biotechnology and pharmaceutical industry, Haberman Associates would like to hear from you. If you are in a biotech or pharmaceutical company, and would like a 15-20-minute, no-obligation telephone discussion of issues raised by this or other blog articles, or an initial one-to-one consultation on an issue that is key to your company’s success, please contact us by phone or e-mail. We also welcome your comments on this or any other article on this blog.
